Abstract
Background
Growing evidence suggests that multiparameter flow cytometric (MFC) measurable residual disease (MRD) assessment in acute myeloid leukemia (AML) should be used for risk stratification prior to allogeneic hematopoietic stem cell transplantation (alloHCT). We investigated the significance of MFC-MRD status in AML patients treated with myeloablative conditioning (MAC) alloHCT at transplant centers within the Polish Adult Leukemia Group (PALG).
Methods
All consecutive patients who underwent MAC alloHCT from HLA identical siblings (SD), family haploidentical donor, matched (10/10) unrelated donor (MUD), or mismatched (9/10) unrelated donor (mMUD) between January 2014 and May 2022 and for whom pre-transplant MFC-MRD was assessed were included in the analysis. MFC-MRD assessment has been routinely performed on bone marrow aspirates obtained before alloHCT since 2014 or as part of the PALG-AML1/2016 study protocol (NCT03257241) since 2018. Flow cytometry laboratory protocols were modified and unified by the cooperating laboratories according to the European LeukemiaNet recommendations in 2018. Residual disease at a level ≥0.1% was considered MRD(+).
Results
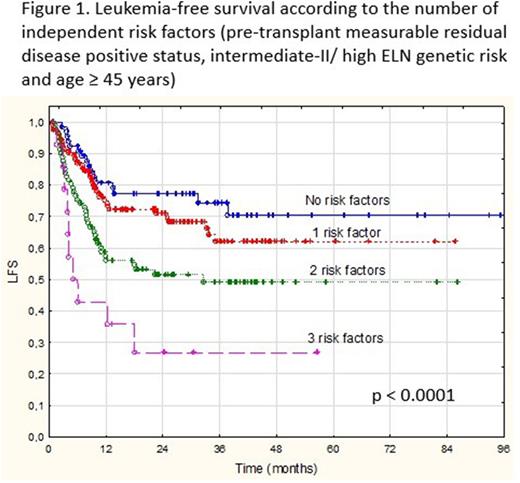
We analyzed 302 adult patients (median age 44 years, range 18-68) with AML in first (88%) or subsequent CR (12%) after intensive chemotherapy, including 81 patients (27%) transplanted between 2014-2017 and 221 (73%) transplanted between 2018-2022. Positive MRD status before alloHCT was detected in 143/302 pts (47%). The European LeukemiaNet (ELN) cytogenetic/genetic risk (Dohner 2010) was available in 290 of the 302 patients and was intermediate-II (Int-II)/high in 22%. Patients received MAC based on i.v. busulfan (84%), treosulfan (1%) or total body irradiation (15%) and graft from either SD (29%), haploidentical donor (9%), MUD (55%), or mMUD (7%). GvHD prophylaxis consisted of calcineurin inhibitor (CNI) combined with MTX (with or without ATG) or was based on post-transplant cyclophosphamide in transplants from haploidentical donor or mMUD. After the median follow-up of 30 months (range, 1-97), the 3-year LFS and OS for MRD(-) vs MRD(+) patients was 69% vs 54% (log-rank p=0.007), and 77% vs 62% (log-rank p=0.013), respectively. In multivariate Cox model, the independent adverse prognostic factors for LFS were MRD positive status (HR 1.53, 95%CI 1.03-2.25; p=0.0332), Int-II/high ELN risk (HR 1.87, 95%CI 1.24-2.84; p=0.0031), and age ≥ 45 years (HR 1.65, 95%CI 1.12-2.45; p=0.011). The same factors independently influenced OS [HR 1.59, 95%CI 1.01-2.48; p=0.0411; HR 1.67, 95%CI 1.03-2.71; p=0.0363, and HR 1.79, 95%CI 1.42-2.79; p=0.0109, respectively]. When the patients were classified according to the number of identified independent risk factors, the 3-year LFS and OS for patients with 0, 1, 2 and 3 risk factors was 75%, 68%, 51%, and 27% (p<0.0001) (Figure 1), and 81%, 77%, 57%, and 44%, respectively (p=0.0006).
Conclusions
Our findings confirm that pre-transplant residual disease at a ≥0.1% level assessed by MFC is an independent poor prognostic factor for both LFS and OS in AML patients treated with MAC alloHCT. As predicted, outcomes after MAC alloHCT were also independently influenced by ELN cytogenetic/genetic risk and age.
Disclosures
Czyz:Pfizer: Honoraria; Amgen: Honoraria; Takeda: Honoraria. Bieniaszewska:Celgene/BMS: Honoraria; Pfizer: Honoraria. Zaucha:Janssen: Honoraria; Abbvie: Honoraria, Membership on an entity's Board of Directors or advisory committees, Other: Consultancy; Gilead: Honoraria; Novartis: Honoraria; BMS: Research Funding; Abbvie: Honoraria; Takeda: Honoraria; Roche: Honoraria. Sobas:Celgene/BMS: Honoraria; Novartis: Honoraria. Wróbel:Celgen/BMS: Honoraria; Abbvie: Honoraria; Takeda: Honoraria; Amgen: Honoraria, Research Funding; Novartis: Honoraria; GSK: Honoraria; Beigene: Honoraria; Gilead: Honoraria; Janssen: Honoraria; Roche: Honoraria, Research Funding. Gil:Janssen: Honoraria; Pfizer: Honoraria; Novartis: Honoraria; Celgene/BMS: Honoraria; Abbvie: Honoraria; Astellas: Honoraria; Gilead: Honoraria. Helbig:Novartis: Honoraria. Mensah-Glanowska:swiss pharma: Honoraria. Pluta:Angelini: Honoraria; Astellas: Honoraria; Celgen/BMS: Honoraria; Novartis: Honoraria; Swixx Biopharma: Honoraria, Research Funding. Patkowska:Bristol Myers: Other: Support for attending meetings and/or travel; Pfizer: Other: Support for attending meetings and/or travel; Astellas Pharma: Honoraria, Other: Support for attending meetings and/or travel; Angelini Pharma: Honoraria, Other: Support for attending meetings and/or travel; Squibb: Other: Support for attending meetings and/or travel; KCR US, Inc: Consultancy; Amgen: Honoraria; Novartis: Honoraria, Other: Support for attending meetings and/or travel; Servier: Honoraria, Other: Support for attending meetings and/or travel; Jazz Pharmaceuticals: Other: Support for attending meetings and/or travel. Desai:Janssen Research: Research Funding; Takeda, Bristol Myers Squibb, Agios: Consultancy, Membership on an entity's Board of Directors or advisory committees. Ritchie:Incyte: Consultancy; Takeda: Consultancy; Novartis: Consultancy; Jazz: Consultancy; Celgene: Consultancy; Pfizer: Consultancy. Guzman:Seq RX: Current holder of stock options in a privately-held company; Samus Therapeutics: Other: Inventor on licensed IP; BridgeMedicines: Research Funding. Roboz:Clovis Oncology: Other: Travel and accommodation expenses; MedImmune: Consultancy, Research Funding; Novartis: Consultancy, Other: Travel and accommodation expenses, Research Funding; Amphivena Therapeutics: Other: Travel and accommodation expenses, Research Funding; Celltrion: Consultancy, Other: Travel and accommodation expenses; Genentech/Roche: Consultancy, Other: Travel and accommodation expenses; Sandoz: Consultancy, Other: Travel and accommodation expenses; MEI Pharma: Consultancy, Research Funding; Pfizer: Consultancy, Honoraria, Other: Travel and accommodation expenses; Mesoblast: Consultancy; Otsuka: Consultancy; Onconova Therapeutics: Research Funding; Daiichi Sankyo: Consultancy; Jazz: Consultancy, Other: travel; Astex Pharmaceuticals: Consultancy, Other: Travel and Accommodation expenses, Research Funding; Array BioPharma: Other: Travel and accommodation expenses; Mofitt Cancer Center: Research Funding; Agios: Other: travel, Research Funding; Amgen: Consultancy, Other: travel; Takeda: Consultancy; Karyopharm Therapeutics: Research Funding; CTI: Research Funding; Sunesis Pharmaceuticals: Other: Travel and accommodation expenses, Research Funding; Eisai: Other: Travel and accommodation expenses; Bayer: Consultancy, Other: Travel and accommodation expenses; Jasper Therapeutics: Consultancy; Janssen: Consultancy, Other: travel and accommodation expenses, Research Funding; GlaxoSmithKline: Consultancy; Celgene: Consultancy, Other: travel and accommodation expenses, Research Funding; Bristol Myers Squibb: Consultancy; Astellas: Consultancy; Amgen: Consultancy; Agios: Consultancy, Research Funding; AbbVie: Consultancy, Other: travel and accommodations, Research Funding; Actinium: Consultancy; Helsinn Therapeutics: Consultancy; Roche: Consultancy; Bristol Myers Squibb: Consultancy; Tensha Therapeutics: Research Funding. Wierzbowska:Novartis: Honoraria; AbbVie: Honoraria; Astellas: Honoraria; Janssen: Honoraria; Gilead: Honoraria; Celgene/BMS: Honoraria; Swixx Biopharma: Honoraria, Research Funding; Servier: Honoraria.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal